About the case
- Therapeutic area: Oncology
- Phase I — III
- Impact: Informing
Principal question
In absence of clear dose-exposure relationships, are there other means to optimize CAR-T cell exposure?
- The main challenge was that although positive exposure-response relationships have been identified for CAR-T cell therapy, traditional dose-exposure relationships do not apply.
- The main impact was the identification of potential MIDD applications for the development of CAR-T cell therapies.
- Pharmetheus identified i) actionable variables which can be modified to optimize exposure; ii) suitable mathematical models to support such optimization processes.
- The method included a review of relevant literature and published computational CAR-T cell models.
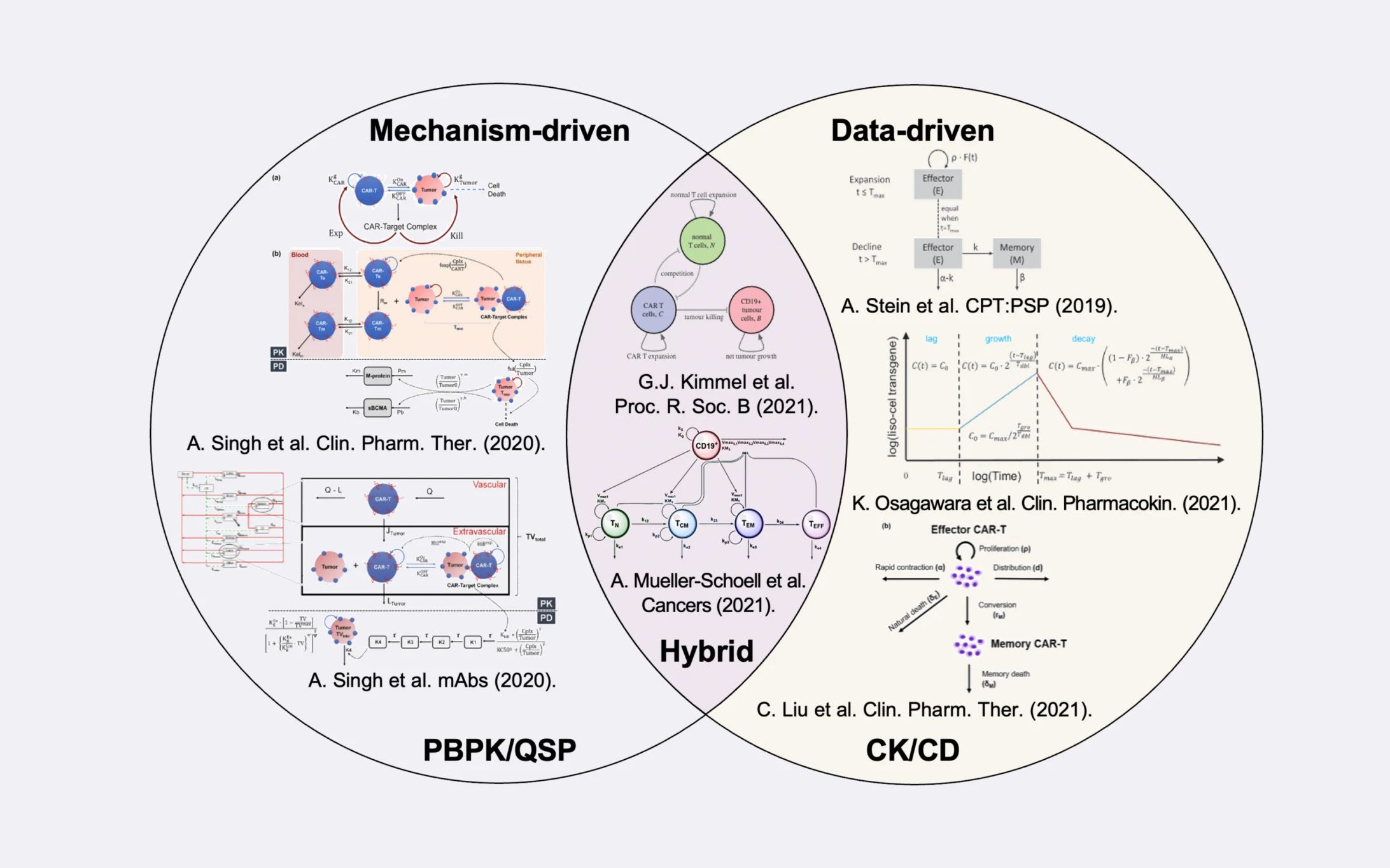
Selection of computational CAR-T cell models that can be used in a model-informed drug development strategy, grouped by the applied modeling approach.
Drug discovery
- Optimization of the CAR affinity
- Selection and optimization of the CAR hinge/spacer domain
- Selection of the CAR co-stimulatory domain(s)
- Selection and optimization of CAR element combinations
Drug development
- Stimulation of cells during the ex vivo expansion
- Optimization of the duration of the ex vivo expansion
- Cytokines in the ex vivo expansion medium and their concentrations
Clinical practice
- Selection of patients who might benefit from bridging therapy and optimization of bridging treatment
- Optimization of preconditioning lymphodepleting chemotherapy
- Combination of CAR-T cells with other treatments
Case Studies
Our case studies showcase our work within all phases of drug development. Each case clearly states the challenges and customer needs, and the services and solutions we provided.