Poster: Model-Based Bioequivalence (MBBE) methods open new doors for generics
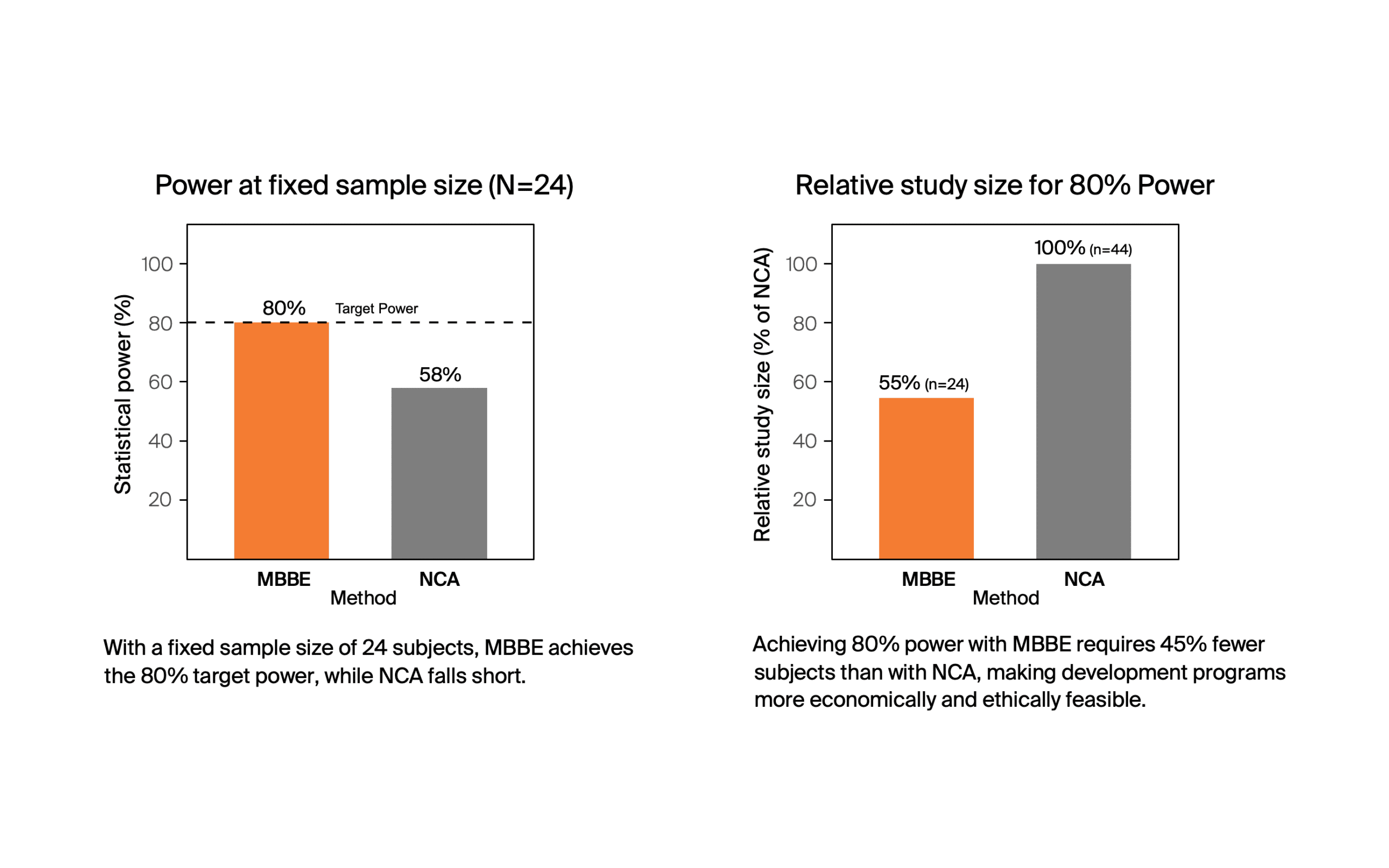
In this example, with a crossover design and moderate variability — where NCA is usually applied, MBBE reduced the required sample size by 45%. MBBE can lower barriers and improve efficiency for bioequivalence studies in general. It can make generics development programs for complex products economically viable, which improves pharmacoequity by making treatments more accessible to patients.
Presented by
Authors
Henrik Bjugård Nyberg a, Andrew C Hooker a, b, Martin Bergstrand a, Marylore Chenel a,
E Niclas Jonsson a
a Pharmetheus AB, Sweden,
b Uppsala University, Sweden
Background
Traditional bioequivalence (BE) via Non-Compartmental Analysis (NCA) has low power and requires prohibitive sample sizes for several classes of drugs, creating barriers to affordable generics. Model-Based Bioequivalence (MBBE) methods can increase the power significantly, while controlling Type I error.
- The objective was to demonstrate the improved power with an MBBE approach compared to standard NCA methodology.
Methods
A 2-way crossover study of a moderately variable oral drug (~50% IIV, ~15% IOV) was simulated 500 times with 24 subjects and 10 samples per subject and analyzed using MBBE and NCA + TOST. Sample size required for 80% statistical power was calculated.
- NCA: NCA and power via NCAPPC__ and powerTOST__ R packages
- MBBE: Single-model method with Sampling Importance Resampling (SIR) uncertainty
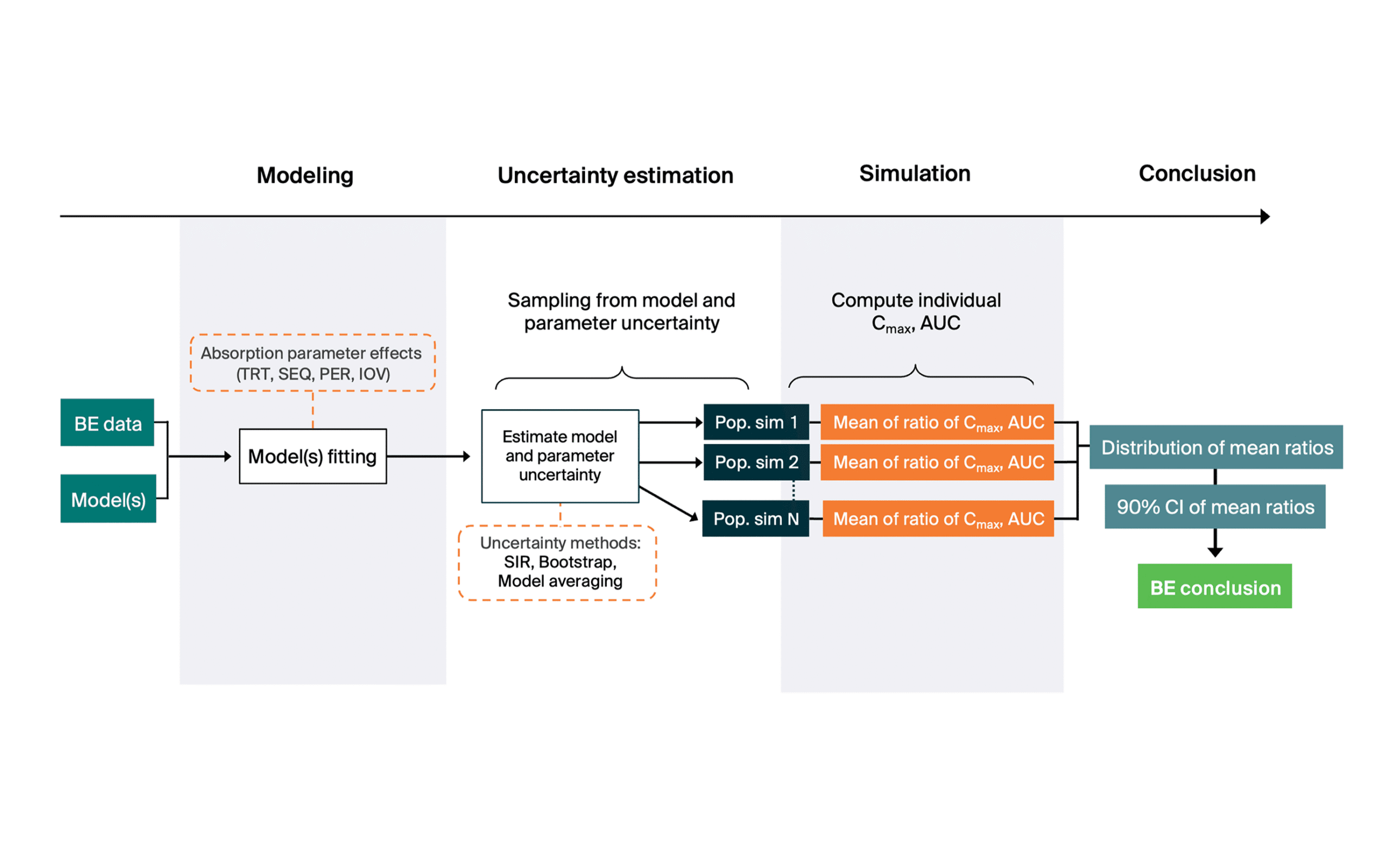
Results
MBBE significantly reduced the required sample size compared to NCA while maintaining target power.
- N for 80% Power – MBBE: 24, NCA: 44
- Power at N=24 — MBBE: 80%, NCA: 58%
Extensions
This is a simple oral formulation example with rich sampling. The impact of MBBE can be much greater in cases that are less well-suited to NCA, such as,
- parallel designs for generic LAIs
- highly variable drugs
- ophthalmics
MBBE may also enable new concepts, e.g.
- new designs (e.g., switch study for LAI instead of steady-state)
- model-based calculation of BE limits
- alternative metrics to AUC and Cmax
Publications
Recommended reading on bioequivalence
Development and comparison of model-integrated evidence approaches for bioequivalence studies with pharmacokinetic end points
Xiaomei Chen, Henrik B Nyberg, Mark Donnelly, Liang Zhao, Lanyan Fang, Mats O Karlsson, Andrew C Hooker
Read the full publication
Evaluation of model-integrated evidence approaches for pharmacokinetic bioequivalence studies using model averaging methods
Henrik B Nyberg, Xiaomei Chen, Mark Donnelly, Lanyan Fang, Liang Zhao, Mats O Karlsson, Andrew C Hooker
New Model–Based Bioequivalence Statistical Approaches for Pharmacokinetic Studies with Sparse Sampling
Florence Loingeville, Julie Bertrand, Thu Thuy Nguyen, Satish Sharan, Kairui Feng, Wanjie Sun, Jing Han, Stella Grosser, Liang Zhao, Lanyan Fang, Kathrin Möllenhoff, Holger Dette, France Mentré.
